Organic Synthesis and Catalysis
Our research philosophy is that the methods we develop should be user-friendly, experimentally trivial, environmentally friendly, and economically sound while providing access to otherwise difficult targets of structural and biological significance.
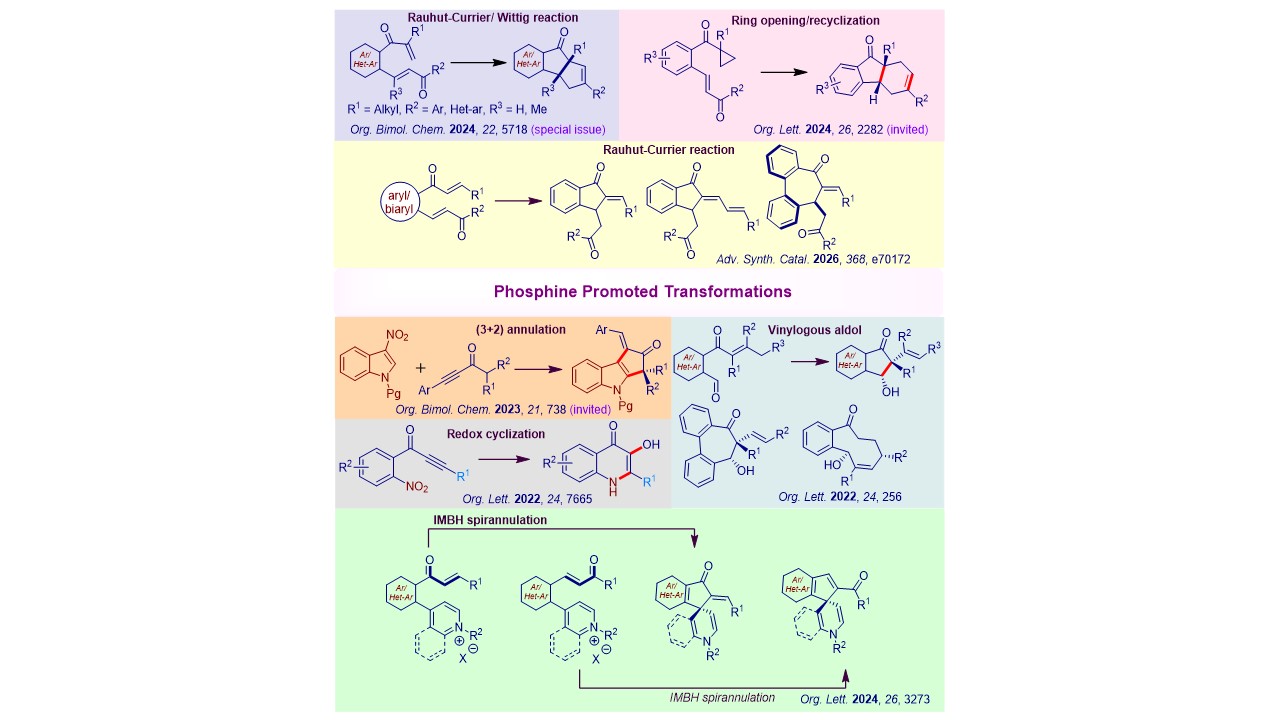
1. Organocatalysis with phosphines We routinely pursue metal-free chemistry with nucleophilic trivalent organophosphines. Some key contributions to this area include the development of asymmetric intramolecular Morita-Baylis-Hillman reaction and its variants, phosphine-promoted reductive- and vinylogous aldol reactions, phosphine-catalyzed intramolecular hydroalkylation, and hydroacylation chemistry, etc.
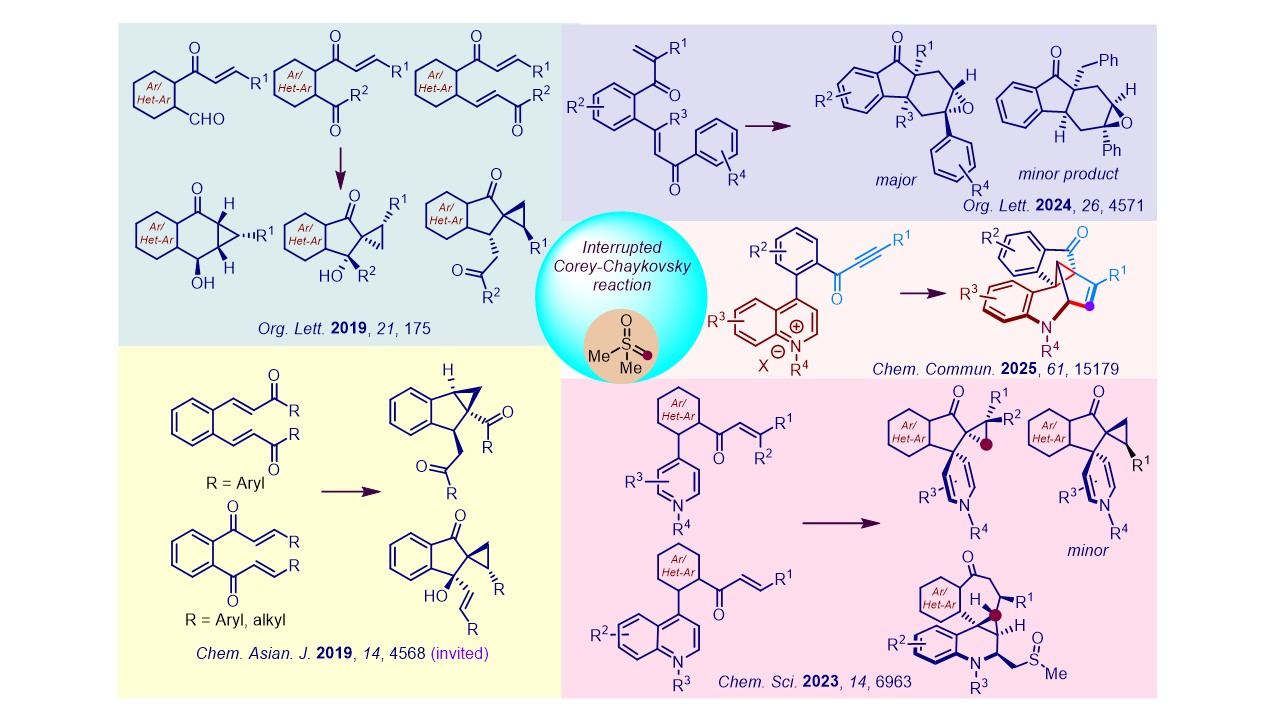
2. Interrrupted Corey-Chaykovsky reactions Corey-Chaykovsky reagent is traditionally employed to synthesize cyclopropanes, epoxides, and aziridines from the respective olefins, carbonyls, and imines. We made a serendipitous entry into this field and articulated several one-pot cascade transformations employing sulfur ylides as reagents, which we named as "interrupted Corey-Chaykovsky reactions" that provided facile access to unprecedented privileged moieties.
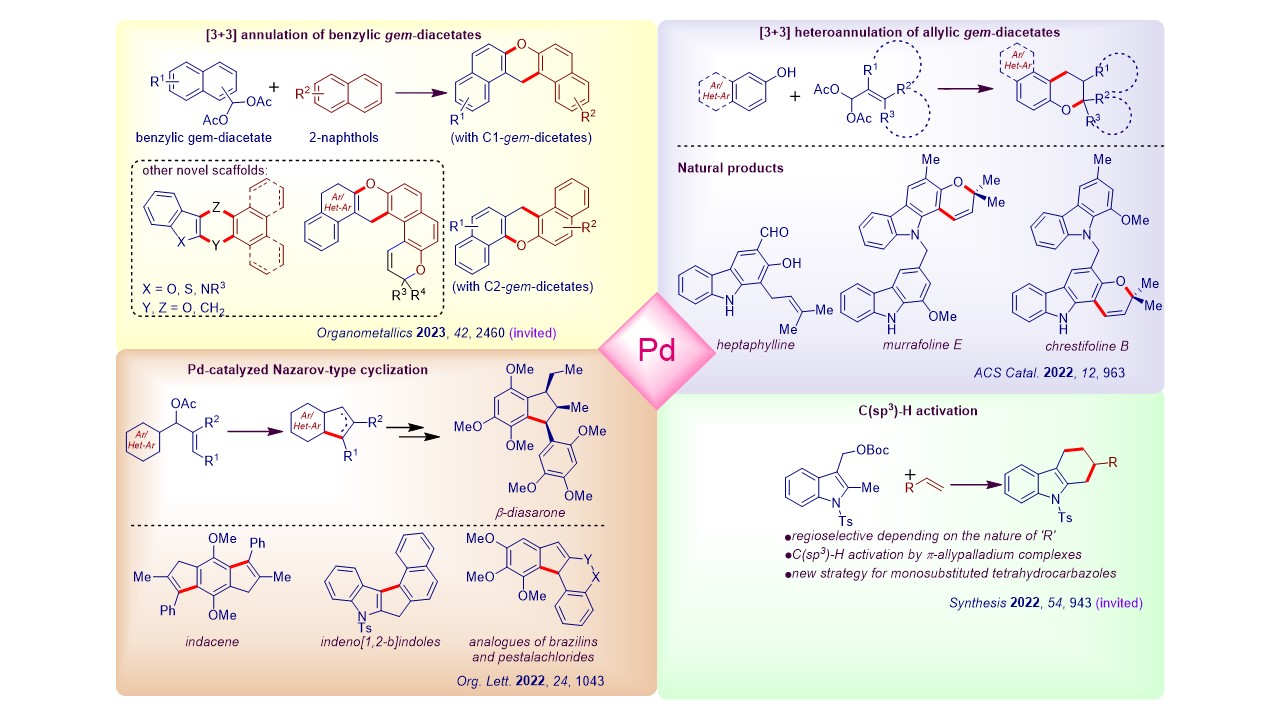
3. Pd-catalyzed annulative allylic alkylation chemistry Our group actively works on developing novel Pd-catalyzed cycloisomerization reactions. We have showcased the utility of these methods in synthesizing many bioactive natural products. We have also assembled several intricate scaffolds through these strategies, which have relevance in medicinal chemistry and materials science.